 The concept of sigma bonding is extended to describe bonding interactions involving overlap of a single lobe of one orbital with a single lobe of another. Sigma bonds are obtained by head-on overlapping of atomic orbitals. A double bond has one sigma plus one pi bond, and a triple bond has one sigma plus two pi bonds. Typically, a single bond is a sigma bond while a multiple bond is composed of one sigma bond together with pi or other bonds. When viewed down the bond axis, a σ MO has a circular symmetry, hence resembling a similarly sounding "s" atomic orbital. Sigma bonds are the strongest type of covalent bonds due to the direct overlap of orbitals, and the electrons in these bonds are sometimes referred to as sigma electrons. The corresponding antibonding, or σ* orbital, is defined by the presence of one nodal plane between the two bonded atoms. The extent of this mixing (or hybridization or blending) depends on the relative energies of the MOs of like symmetry.ġsσ* antibonding molecular orbital in H 2 with nodal planeįor homodiatomics ( homonuclear diatomic molecules), bonding σ orbitals have no nodal planes at which the wavefunction is zero, either between the bonded atoms or passing through the bonded atoms. As a practical consequence of this mixing of diatomic molecules, the wavefunctions s+s and p z+p z molecular orbitals become blended. 
Quantum theory also indicates that molecular orbitals (MO) of identical symmetry actually mix or hybridize. By this definition, common forms of sigma bonds are s+s, p z+p z, s+p z and d z 2+d z 2 (where z is defined as the axis of the bond or the internuclear axis). 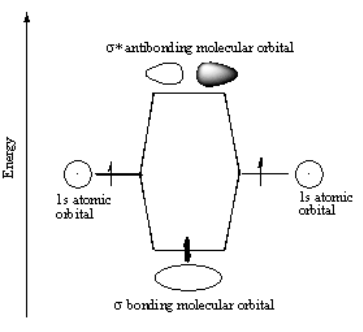
In this formal approach, a σ-bond is symmetrical with respect to rotation about the bond axis. Sigma bonding is most simply defined for diatomic molecules using the language and tools of symmetry groups. They are formed by head-on overlapping between atomic orbitals. In chemistry, sigma bonds ( σ bonds) are the strongest type of covalent chemical bond. Covalent chemical bond σ bond between two atoms: localization of electron density 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
